Expressing milk antenatally has been mentioned in medical journals since the 1940s and 50s, but has only become common in maternity care in recent years. The practice has had a resurgence, probably due to a few factors including the World Health Organization developing the Baby Friendly Hospital Initiative in 1991 to promote, support and protect breastfeeding. As a result, health organisations have refocused on the benefits of breastfeeding for maternal and infant health, and now have breastfeeding targets to achieve during women’s postpartum hospital stay.
Women with diabetes during pregnancy make up between seven and 14 per cent of the pregnant population. These women and their infants are at increased risk of complications during pregnancy. After the birth, the babies are at higher risk of hypoglycaemia than babies of women who do not have diabetes in pregnancy. These women are at increased risk of delayed lactogenesis II (breastmilk ‘coming in’ more than 72 hours postpartum). These complications and the delay in lactogenesis II may lead to mother-infant separation, early formula supplementation or shorter breastfeeding duration.1 Despite the potential challenges, pregnant women with diabetes are actively encouraged to breastfeed because of the positive metabolic effects breastfeeding confers and the increased chance of delaying the onset of type 2 diabetes for women with gestational diabetes mellitus (GDM).
Emerging practice of antenatal expressing despite little or no evidence
The purpose of expressing before the birth is to have an adequate volume of breastmilk available for supplementary feeding to treat neonatal hypoglycaemia, hopefully keep mother and baby together, promote earlier onset of lactogenesis II, and, possibly, increase women’s breastfeeding confidence. Other reasons women might express antenatally are the ‘just in case’ scenario, or because of previous poor breastfeeding experiences. However, until recently, there has been very little evidence to support the practice other than case studies and two pilot studies.2 3
The topic of antenatal expressing became controversial, with some clinicians calling for the practice to cease until safety and efficacy were established. Others were of the belief that women have breastfed during pregnancy so antenatal expressing would not cause any harm. A 2014 Cochrane review4 concluded that there was ‘no high level systematic evidence to inform the safety and efficacy of the practice’.

Marie and her baby. Marie expressed breastmilk antenatally.
Diabetes and antenatal milk expressing (DAME): a randomised controlled trial
In order to establish evidence for this practice, the DAME trial was conducted in Melbourne from 2011 to 2015. The trial was a multicentre, two-group, unblinded randomised controlled trial involving six hospitals in Victoria, funded by the National Health and Medical Research Council. Six hundred and thirty-five eligible low-risk women with diabetes during pregnancy (93 per cent GDM) were randomised to hand express twice a day from 36 weeks or to have normal care (no expressing). Given this was the first trial of antenatal expressing in the world and women with diabetes have an increased degree of pregnancy risk, careful identification of pre-existing and pregnancy complications were considered and women needed to have a reassuring cardiotocograph prior to randomisation.
Women allocated to the expressing arm of the trial were taught how to hand express. They were given a diary to document the volume expressed and any other thoughts or feelings about expressing. The milk was frozen and stored at home in syringes. The frozen breastmilk was then brought to hospital and stored in the postnatal ward freezer ready for use if required. Follow up was by comprehensive data collection from medical records at birth and during telephone interview at two weeks and three months.

Edel and her baby. Edel was a participant in the DAME trial.
Summary of results
The DAME trial was published in June 20175 and found no difference in the proportion of infants admitted to the NICU (primary outcome): 14.5 per cent (n=46) in the intervention (advised to express) arm compared with 140 per cent (n=44) in standard care (Adj. RR 1.06; 95% CI 0.66, 1.46). There was no difference in the mean gestational age at birth (mean difference 0.05; 95% CI–0.10, 0.21). There was moderate evidence of association between allocation to antenatal expressing during pregnancy and the proportion of infants receiving exclusive breastmilk in the first 24 hours of life (Adj. RR 1.15; 95% CI 1.02, 1.28), and some evidence of an association with receiving exclusive breastmilk during the initial hospital stay (Adj. RR 1.16; 95% CI 0.99, 1.33). Women expressed many times before birth, with a median of 20 times (range 1, 59; IQR 9, 33). The median total volume expressed was 5.5mL (range 0, 905; IQR 0.25, 22), which was significantly less than many clinicians had expected.
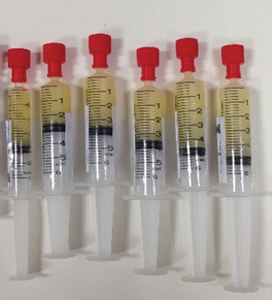
Syringes of antenatal breastmilk.
Interpreting the results
This trial was limited to a low-risk subset of women with diabetes during pregnancy. The results should not be extrapolated to high-risk groups, as the results may be different. Clinicians are asking, given the apparent safety for low-risk women with diabetes, should the practice of expressing antenatally be advised for all low-risk women, not just those with diabetes? Until there is further research in this field, we should be cautious about interrupting highly sensitive normal postpartum breastfeeding in the absence of medical indications (for example, neonatal hypoglycaemia). Additionally, given that most women expressed low volumes (median 5.5mL total), some women, especially primiparous women, may be concerned about their ability to produce adequate milk volumes after their baby is born.
Others argue that the act of antenatal expressing may in fact increase confidence with breastfeeding, as women will feel they have already mastered the valuable skill of hand expressing regardless of the volume of milk expressed. These are important questions for consideration and further research is required before routine advising of antenatal expressing.
Where to from here?
Given the lack of evidence-based information, local and international dissemination of the findings has been undertaken to better inform clinicians and breastfeeding peer support groups. The DAME trial research group produced an article for the wider population in The Conversation, called ‘Health Check: is it safe to express milk before giving birth?’6 Members of the research group have also worked with the Australian Breastfeeding Association to update their online information for women.
In order to reflect best practice on antenatal expressing, members of the research group are working with the three tertiary maternity hospitals in Victoria to develop clinical practice guidelines and a fact sheet for women with diabetes during pregnancy. These guidelines will also provide consistent criteria and processes among maternity providers.
With hard work and great collaboration, we are moving towards better information for clinicians and women about antenatal expressing during pregnancy and have identified areas for further research.
References
- De Bortoli J, Amir LH. Is onset of lactation delayed in women with diabetes in pregnancy? A systematic review. Diabet Med. 2016;33(1):17-24.
- Soltani H, Scott AM. Antenatal breast expression in women with diabetes: outcomes from a retrospective cohort study. Int Breastfeed J. 2012;7:18.
- Forster DA, McEgan K, Ford R, Moorhead A, Opie G, Walker S, et al. Diabetes and antenatal milk expressing: a pilot project to inform the development of a randomised controlled trial. Midwifery. 2011;27(2):209-14.
- East CE, Dolan WJ, Forster DA. Antenatal breast milk expression by women with diabetes for improving infant outcomes. Cochrane Database Syst Rev. 2014(7):CD010408.
- Forster DA, Moorhead AM, Jacobs SE, Davis PG, Walker SP, McEgan KM, et al. Advising women with diabetes in pregnancy to express breastmilk in late pregnancy (Diabetes and Antenatal Milk Expressing [DAME]): a multicentre, unblinded, randomised controlled trial. Lancet. 2017;389(10085):2204-13.
- Amir L, Moorhead A, Forster D, Walker S. Health Check: is it safe to express milk before giving birth? The Conversation. 2017 18.01.2018. Available from: https://theconversation.com/health-check-is-it-safe-to-express-milk-before-giving-birth-78399.







Leave a Reply